Smart Codes make it easy
Screening for cervical cancer and STIs, simplified
Delivering the care your patients need should not be complicated. Smart Codes from Quest are based on guidelines and use a patient’s age and STI risk factors to determine which combination of HPV and select STI screening, along with an image-guided Pap, should be performed.


Trust Smart Codes to order appropriate guidelines-based screening for your patients
Smart Codes follow the current guidelines1-5 and help you order the appropriate screening with the right codes to deliver optimal patient care and improve practice workflow.
| Tests included with Smart Code 91384 Panel components can be ordered separately |
|
|---|---|
| Ages (years) | 91384–Image-Guided Papa with Age-Based Screening Protocols |
| 21-24 |
|
| 25-29 |
|
| 30-65 |
|
| Tests included with Smart Code 91384 Panel components can be ordered separately |
|---|
| 91384–Image-Guided Papa with Age-Based Screening Protocols |
|
AGES (YEARS) 21-24
|
|
AGES (YEARS) 25-29
|
|
AGES (YEARS) 30-65
|
| Tests included with Smart Code 91386 and 91385 Panel components can be ordered separately |
||
|---|---|---|
| Ages (years) | 91386–Image-guided Papa with Age-Based Screening, with CT/NG, Trichomonas | 91385–Image-guided Papa with Age-Based Screening, with CT/NG |
| 21-24 | Same as 91384, plus Trichomonas (guidelines recommend routine screening of CT/NG in women ages 21-244) | Same as 91384 (guidelines recommend routine screening of CT/NG in women ages 21-244) |
| 25-29 | Same as 91384, plus CT/NG and Trichomonas | Same as 91384, plus CT/NG |
| 30-65 | Same as 91384, plus CT/NG and Trichomonas | Same as 91384, plus CT/NG |
| Tests included with Smart Code 91386 and 91385 Panel components can be ordered separately |
|---|
| 91386–Image-guided Papa with Age-Based Screening, with CT/NG, Trichomonas |
|
AGES (YEARS) 21-24 Same as 91384, plus trichomonas (guidelines recommend routine screening of CT/NG in women ages 21-244) |
|
AGES (YEARS) 25-29 Same as 91384, plus CT/NG and Trichomonas |
|
AGES (YEARS) 30-65 Same as 91384, plus CT/NG and Trichomonas |
| Tests included with Smart Code 91385 |
|---|
| 91385–Image-guided Papa with Age-Based Screening, with CT/NG |
|
AGES (YEARS) 21-24 Same as 91384 (guidelines recommend routine screening of CT/NG in women ages 21-244) |
|
AGES (YEARS) 25-29 Same as 91384, plus CT/NG |
|
AGES (YEARS) 30-65 Same as 91384, plus CT/NG |
HPV mRNA assay (90887); Chlamydia trachomatis (CT)/Neisseria gonorrhoeae (NG) RNA, TMA (11363); HPV mRNA Genotypes 16, 18/45 (90942); Trichomonas vaginalis, Qualitative TMA, Pap Vial (90521)
Quest Diagnostics Smart Codes may not represent all of the possible screening options in published guidelines. Please contact your Quest Diagnostics representative for information on additional Pap and HPV testing options from Quest.
For all 3 Smart Codes, if a patient is over the age of 65: Screening should be discontinued if patient has had adequate negative prior screening resultsb and no history of CIN 2+.
Recommend continuing age-based screening for >=20 years in those patients with a history of CIN 2, CIN 3, or adenocarcinoma in situ. Adequate negative prior screening results is defined as 3 consecutive negative Paps or 2 consecutive negative co-tests within the past 10 years, with the most recent test occurring within the past 5 years.1-3
aACOG recommendations do not specify a particular type of Pap test. Both imaged and nonimaged Pap tests are acceptable under the recommendations. Information on nonimaged Pap tests can be found at QuestDiagnostics.com/TestDirectory
bDefined as 3 consecutive negative Paps or 2 consecutive negative co-tests within the past 10 years, with the most recent test occurring within the past 5 years.
Count on Quest for tests that deliver highly accurate results
Screening for cervical cancer has dramatically helped reduce its incidence and mortality, and co-testing with Pap and HPV together offer superior detection of risk versus either test alone.6
Count on Quest for tests that deliver highly accurate results
Screening for cervical cancer has dramatically helped reduce its incidence and mortality, and co-testing with Pap and HPV together offer superior detection of risk versus either test alone.6
ThinPrep® is an image-guided Pap test using a liquid-based cytology collection system that delivers:
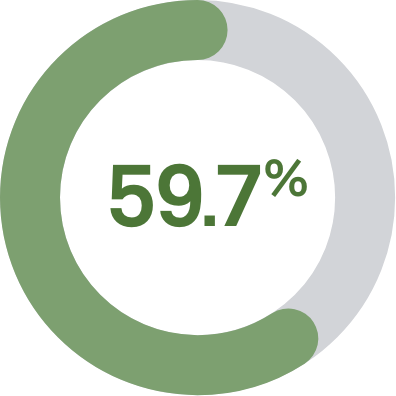
The Aptima® HPV mRNA assay detects the presence and activity of HPV infection and is shown to reduce false positives by:
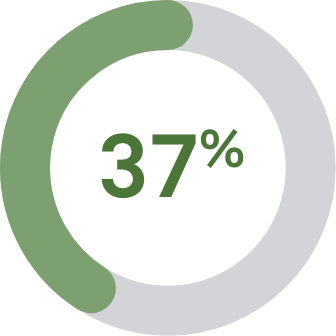
vs Roche cobas® HPV8
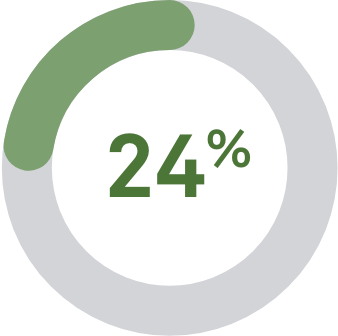
vs digene HC29

vs Roche cobas® HPV8

vs digene HC29
SureSwab® (ATV) assay uses NAAT, the most reliable diagnostic test method, and yields up to:


in the detection of T. vaginalis
Smart Codes simplify the ordering process so you can provide the care she needs
Use Smart Codes today to order the screening your patients need
References:
- Women’s Preventive Services Initiative. Recommendations for well-woman care – a well-woman chart. Published August 26, 2019. Accessed August 11, 2021. https://www.womenspreventivehealth.org/wp-content/uploads/WellWomanChart.pdf
- ACOG. Updated cervical cancer screening guidelines. Updated April 2021. Accessed August 11, 2021. https://www.acog.org/clinical/clinical-guidance/practice-advisory/articles/2021/04/updated-cervical-cancer-screening-guidelines
- US Preventive Services Task Force. Cervical cancer: screening. Published August 21, 2018. Accessed August 11, 2021. https://www.uspreventiveservicestaskforce.org/uspstf/recommendation/cervical-cancer-screening
- CDC. Sexually transmitted infections treatment guidelines, 2021. Reviewed July 22, 2021. Accessed August 11, 2021. https://www.cdc.gov/std/treatment-guidelines/screening-recommendations.htm
- Perkins, RB, Guido, RS, Castle, PE, et al. 2019 ASCCP Risk-based management consensus guidelines for abnormal cervical cancer screening tests and cancer precursors. J Low Genit Tract Dis. 2020;24(RR-02): 102–131. doi:10.1097/LGT.0000000000000525
- Kaufman HW, Alagia DP, Chen Z, et al. Contributions of liquid-based (Papanicolaou) cytology and human papillomavirus testing in co-testing for detection of cervical cancer and precancer in the United States. Am J Clin Pathol. 2020;154(4):510-516. doi:10.1093/ajcp/aqaa074
- ThinPrep® 2000 system package insert. MAN-02624-001 Rev. 004. Hologic, Inc.; 2017.
- Cuzick J, Cadman L, Mesher D, et al. Comparing the performance of six human papillomavirus tests in a screening population. Br J Cancer. 2013;108(4):908-913. doi:10.1038/bjc.2013.22
- Monsonego J, Hudgens MG, Zerat L, et al. Evaluation of oncogenic human papillomavirus RNA and DNA tests with liquid-based cytology in primary cervical cancer screening: the FASE study. Int J Cancer. 2011;129(3):691-701. doi:10.1002/ijc.25726
- Hobbs MM, Seña AC. Modern diagnosis of Trichomonas vaginalis infection. Sex Transm Infect. 2013;89(6):434-438. doi:10.1136/sextrans-2013-051057

Genetic consultation available
Genomic science specialists are available to help with test selection and results interpretation 1.866.GENE.INFO (1.866.436.3463)





